Covid 19: Laboratory findings and patient safety
Patient Safety During the Covid-19 Pandemic
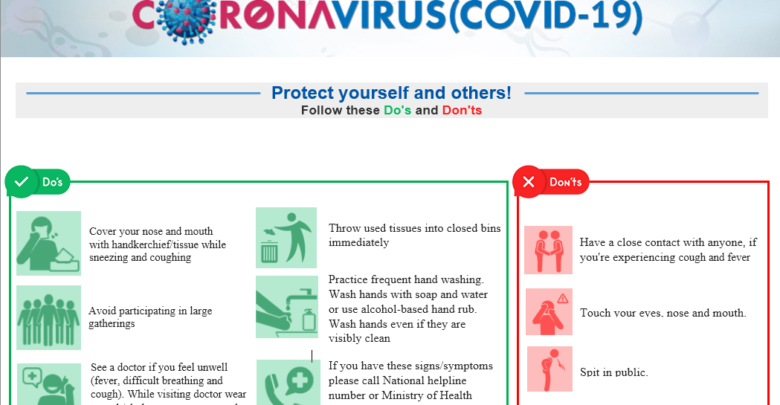
The International Society for Quality in Health Care (ISQua) has published the Patient Safety Recommendations for Covid-19 Epidemic Outbreak (Lessons from the Italian Experience) with their partners in Italy. On the basis of reports and questions forwarded to the Clinical Risk Managers of the Italian Network for Health Safety (INSH) from physicians working on the front line, a series of recommendations have been developed referring to documents and papers published by national institutions (ISS) and Italian and international scientific societies and journals. The document is work in progress and will be subject to updates by all professionals on a continuing basis.
ISQua´s recommendations for diagnosis – Role of medical laboratories
Medical laboratories have an important role in diagnosis and monitoring the patient care as usual. Here are a few key excerpts from the recommendation that emphasize the role of the laboratory.
The adequate specimen for Real Time-Polymerase Chain Reaction (RT-PCR) testing is nasopharyngeal and oropharyngeal sampling. Prefer lower respiratory tract (LRT; expectorated sputum, endotracheal aspirate, or bronchoalveolar lavage) when readily available (for example, in mechanically ventilated patients). Quality of RT-PCR testing is a crucial issue. Both pre-analytical and analytical variables should be carefully considered, and a validation process should be performed according to ISO 15189.
A “COVID-19 profile” should be defined for the rapid order entry of blood tests, including the following tests: blood count, CRP, creatinine, electrolytes, blood glucose, albumin, AST, ALT, LDH, bilirubin, pneumococcal and legionella urinary agents, PT-INR, troponin and procalcitonin.
The most common reported laboratory abnormalities in COVID-19 patients are: Lymphopenia (35-75%), increased CRP (75-93%), LDH (27-92%), ESR (up to 85% of cases), hypoalbuminemia (50-98%) and anemia (41-50%).
The following negative prognostic factors have been reported: leukocytosis, neutrophilia, increased procalcitonin, LDH, AST, ALT, total bilirubin, creatinine, troponin, d-dimer, PT and hypoalbuminemia, lymphopenia. Even thrombocytopenia is associated with severe disease.
Other respiratory infections (legionella, pneumococcus, mycoplasma, chlamydia, other respiratory viruses) should not be forgotten even during epidemics, so look for other pathogens and consider antibiotics. During epidemics it is important to avoid availability bias that means diagnose all infections due to epidemic agents. WHO recommends investigating other pathogens, as co-infections have been reported.
Discover more from MedCircles Ghana
Subscribe to get the latest posts sent to your email.